
Chemistry Class 11 NCERT Solutions Chapter 4 Chemical Bonding and Molecular Structure Part 2
This widget gets the Lewis structure of chemical compounds. Get the free "Lewis Structure Finder" widget for your website, blog, Wordpress, Blogger, or iGoogle. Find more Chemistry widgets in Wolfram|Alpha.

[Punjabi] Draw lewis dot structure of PH3
A Lewis electron dot diagram (or electron dot diagram, or a Lewis diagram, or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The number of dots equals the number of valence electrons in the atom. These dots are arranged to the right and left and above and below the.

PH3 Lewis StructureLewis Structure of PH3 (Phosphorus Trihydride)Draw Lewis Structure for PH3
---- Steps to Write Lewis Structure for compounds like PH3 ---- 1. Find the total valence electrons for the PH3 molecule. 2. Put the least electronegative atom in the center. Note: Hydrogen.

PH3 Lewis Structure How to Draw the Lewis Structure for PH3 YouTube
* Phosphorus requires a full octet of electrons, and brings 5 with it.* Each Hydrogen brings 1 electron* This allows for three single bonds and ONE lone pair.
Ph3 Lewis Structure Shape
Lewis Structure is the pictorial representation of the arrangement of atoms and valence electrons in the molecule. To know the Lewis Structure, we first know the central atom and the arrangement of other atoms. Here for PH3, the phosphorus atom will take the central position as Hydrogen atoms cannot take a central position in the Lewis Structure.

H Lewis Dot Structure
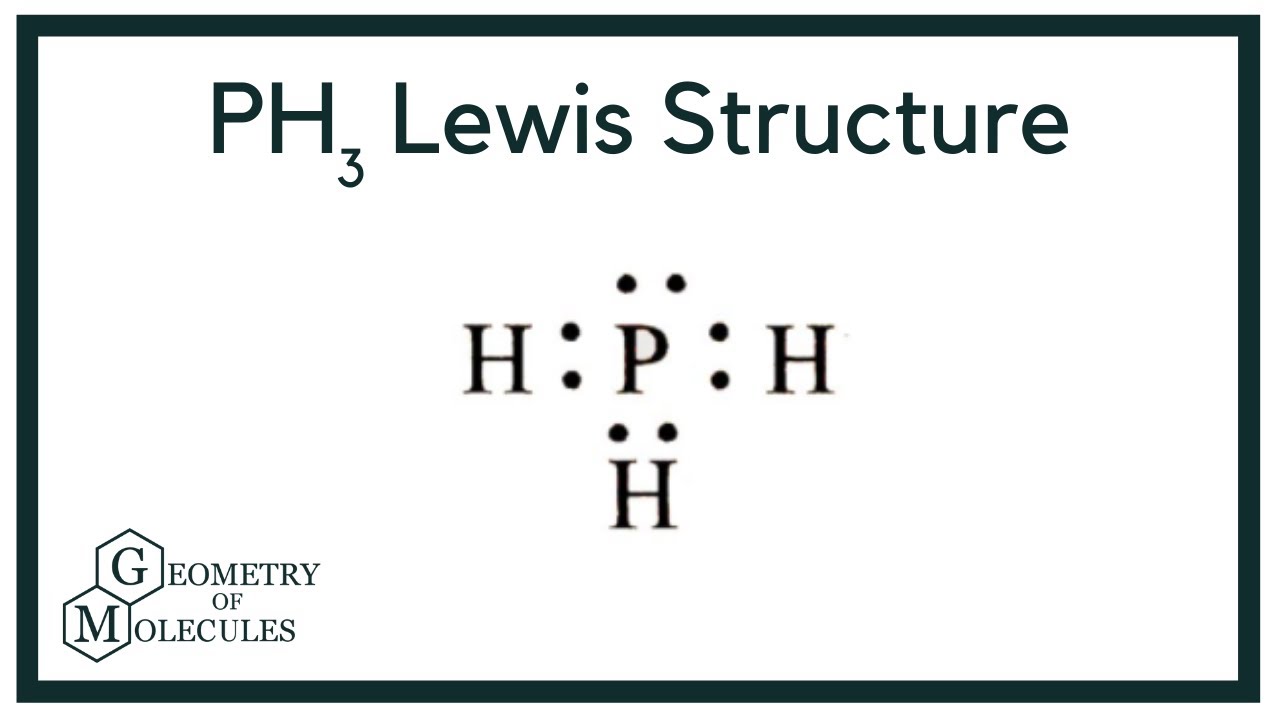
Lewis structure of PH3 contains three single bonds between the Phosphorus (P) atom and each Hydrogen (H) atom. The Phosphorus atom (P) is at the center and it is surrounded by 3 Hydrogen atoms (H). The Phosphorus atom has one lone pair. Let's draw and understand this lewis dot structure step by step.
PH3 Lewis structure YouTube
Shape of PH 3 is trigonal pyramidal. Molecular geometry around phosphorous atom is tetrahedral. Total valence electrons pairs around phosphorous atom is four. In this tutorial we will learn how to draw the lewis structure of PH 3 and determining the shape and molecular geometry of the molecule. PH 3 lewis structure

write the lewis dot structure of PH3 H2O Chemistry Chemical Bonding and Molecular Structure
A dot structure is any representation of atoms/molecules using dots for electrons. And a Lewis diagram (or Lewis structure or Lewis dot structure) is a type of dot structure created by the chemist Gilbert N. Lewis which is most commonly used in chemistry nowadays. There's a slight difference, but they effectively mean the same thing.

Lewis dot structure of PH3 (phosphine)... YouTube
Phosphine (PH3) lewis dot structure, molecular geometry, hybridization, Bond angle, polar or nonpolar Phosphine is a very toxic and dangerous gas, it has a chemical formula of PH 3. It is a colorless gas and has an odor like fish or garlic.

Ph3 Lewis Structure Shape
The PH3 Lewis structure has 8 valence electrons. Remember that hydrogen (H) only needs two valence electrons to have a full outershell. The Lewis structure for PH3 is similar the the structure for NH3 since both P and N are in the same group on the Periodic table. PH3 Lewis Structure - How to Draw the Lewis Structure for PH3 It is helpful if you:
Solved Part A Draw the Lewis structure of PH3 To add lone
From the Lewis molecular structure of PH3, we have seen the phosphorous atom has five valence electrons. During the bonding process, Phosphorous is surrounded by three hydrogen atoms, and each is connected by a single bond. The two remaining electrons form a lone pair.

Draw the Lewis dot structure for PH3 YouTube
We will study the PH3 lewis structure and understand the concept. Some facts about Phosphane. PH3 has a molar mass equal to 33.99 g/mol. It exists as a gas that is colorless and has an odor like that of a rotten fish. The density of PH3 is 1.37 g/L. The melting point of the compound is said to be-132 degrees Celsius and the observed boiling.

Solved What is the correct LewisDot structure for PH3? А. Н
Phosphine PH3 Lewis Dot Structure shadowboy220 1.9K subscribers 49K views 11 years ago Chemistry Lewis Dot Structures A video explanation of how to draw the Lewis Dot Structure for.

How To Draw Electron Dot Diagrams Elementchampionship Jeffcoocctax
The Lewis structure for PH 3 is similar to NH 3. In the PH 3 Lewis structure (and all Lewis structures) hydrogen goes on the outside. Remember, too, that hydrogen only needs two valence electrons to have a full outer shell. In the Lewis structure for PH 3 there are a total of 8 valence electrons. Three pairs will be used in the chemical bonds.

Lewis dot structure covalent bond ph3 Jennifer Reyes, Jenna Kriho Science ShowMe
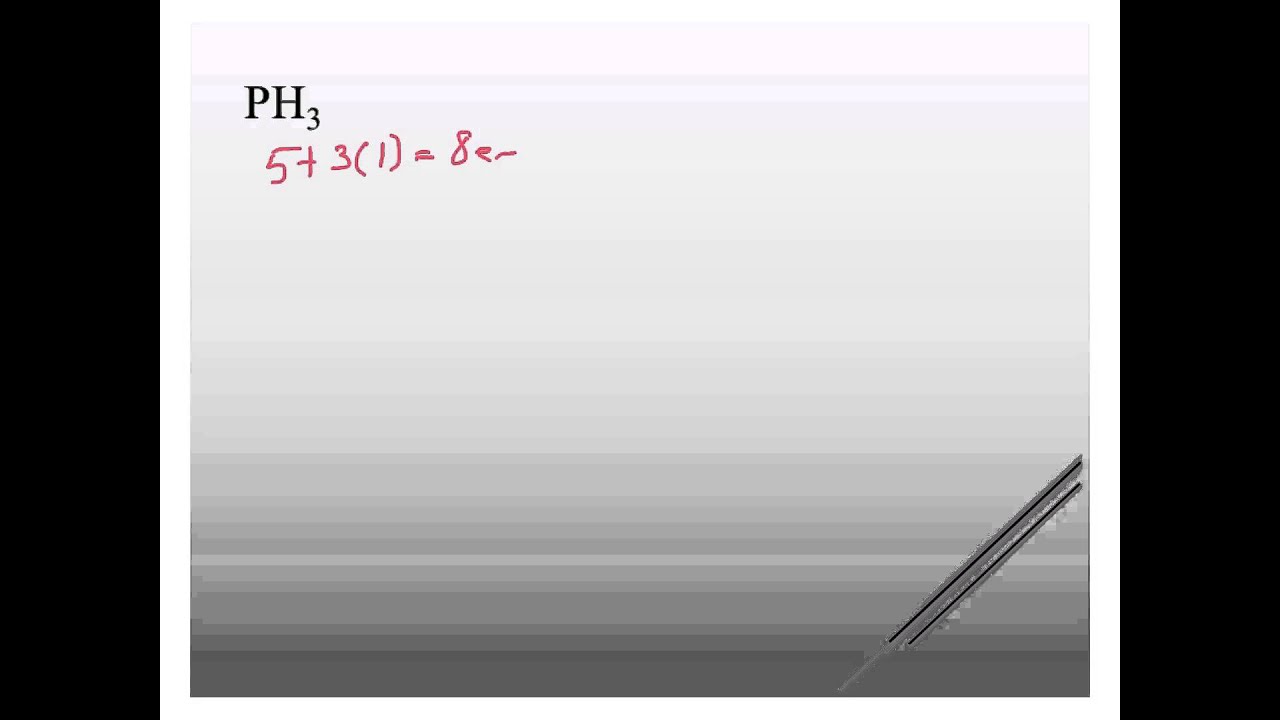
Draw a trial structure by putting electron pairs around every atom until each gets an octet. Count the valence electrons in your trial structure (8). Now, count the valence electrons you actually have available. 1 P + 3 H =. 1×5 + 3×1 = 8. The trial structure has exactly the same number of electrons as we have available.
PH3 Lewis Structure (Phosphine) YouTube
A Lewis symbol consists of an elemental symbol surrounded by one dot for each of its valence electrons: Figure 7.9 shows the Lewis symbols for the elements of the third period of the periodic table. Figure 7.9 Lewis symbols illustrating the number of valence electrons for each element in the third period of the periodic table.